Publications
Filter by type:
High-Content Screening Framework for Academic Early Drug Discovery
Abstract

High-content screening (HCS), a phenotypic-based approach, leverages cellular assays, automated micros-copy, and bioimage analysis to efficiently describe the response of complex biological systems to thousands of individual compounds. Herein, we present a framework for high-throughput academic drug discovery campaigns using HCS methodologies. The workflow covers assay development, screening, data analysis, and hit validation, offering detailed guidance for success.
Unveiling the intricate dynamics of the interplay between triple-negative breast cancer cells and the blood-brain barrier endothelium
Abstract
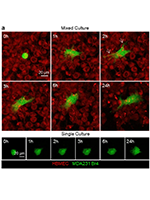
Brain metastases (BM) critically reduce breast cancer (BC) patients’ survival. Extravasation is pivotal for BM development, but the underlying events at the blood-brain barrier (BBB) remain elusive. We aimed to unravel the players and mechanisms governing BC cells (BCCs)-BBB interaction. For that, mixed cultures of human brain microvascular endothelial cells (HBMECs), mimicking the BBB, and brain-tropic triple-negative BCCs (MDA-MB-231 Br4), or non-brain-tropic (MDA-MB-231) or non-metastatic cells (MCF-7) were established. Temporal and spatial analysis of BCCs-BBB interactions (live-cell imaging automated microscopy), and assessments of endothelial-to-mesenchymal transition (EndMT) markers, transcription factors, cytoskeletal proteins, and morphology (immunocytochemistry) were performed. BBB integrity (permeability, transendothelial electrical resistance) and endothelial migration (wound-healing) were also assessed. Our results revealed that contrasting with non-metastatic and non-brain-tropic cells, BCCs quickly developed an invasive, migratory phenotype, characterized by invadopodium formation and reduced roundness. Spatial analysis showed different positioning of BCCs relative to the BBB endothelium over time, with 14% of BCCs transmigrated after 3 h, compromising BBB integrity through endothelial holes, reduced tightness, and increased permeability. Prior to transmigration, alterations in adhesion markers (E-selectin, ICAM-1, CD24, CD34, β3-integrin, Sialyl-Lewis X) were observed. EndMT was also evident by decreased endothelial (β-catenin and pan cytokeratin) and increased mesenchymal (vimentin, neuronal-cadherin, Slug, ZEB1) markers, elongation (RhoA, α-SMA), nuclear deformation, and migratory capacity. Caveolin-1 silencing in HBMEC decreased BCCs transmigration. This study reveals significant BBB phenotypic and structural changes, facilitating both paracellular and transcellular BCCs transmigration. These findings provide advanced understanding of BCCs trafficking across the BBB, aiding strategy development to prevent extravasation and BM.
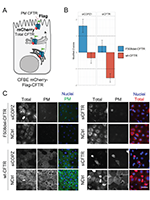
Global Functional Genomics Reveals GRK5 as a Cystic Fibrosis Therapeutic Target Synergistic with Current Modulators
Abstract

Cystic Fibrosis (CF) is a life-shortening disease affecting >160,000 individuals worldwide predominantly with respiratory symptoms. About 80% of individuals with CF have the p.Phe508del variant that causes the CF transmembrane conductance regulator (CFTR) protein to misfold and be targeted for premature degradation by the endoplasmic reticulum (ER) quality control (ERQC), thus preventing its plasma membrane (PM) traffic. Despite the recent approval of a ‘highly effective’ drug rescuing p.Phe508del-CFTR, maximal lung function improvement is ∼14%. To identify global modulators of p.Phe508del traffic, we performed a high-content siRNA microscopy-based screen of >9,000 genes and monitored p.Phe508del-CFTR PM rescue in human airway cells. This primary screen identified 227 p.Phe508del-CFTR traffic regulators, of which 35 could be validated by additional siRNAs. Subsequent mechanistic studies established GRK5 as a robust regulator whose inhibition rescues p.Phe508del-CFTR PM traffic and function in primary and immortalized cells, thus emerging as a novel potential drug target for CF.
Systems Biology and the New Omics
Abstract

While Molecular Biology (a science born in the 1960s by the combined efforts of Biochemistry and Genetics from the discovery that the DNA molecule is the support of genetic information) focuses on characterizing the molecular components of living cells, Systems Biology involves predicting how the collective interactions of the cellular components give rise to the physiology and pathology of a given system (1). Since its beginning around 2000, Systems Biology has thus been revolutionizing the biological sciences, by exploring the concept that “the whole is greater than the sum of the parts”. Advances in Systems Biology have relied very much on the technological developments derived from the Human Genome Project (HGP), the socalled “Omics”, that allow the identification of the total content of a given set of molecules (2). Indeed, the Omics approaches have flourished in the biomedical field and cystic fibrosis (CF) is not an exception. The use of genomics, transcriptomics, proteomics (and interactomics), functional genomics, and also metabolomics have contributed significantly to the understanding of human disease mechanisms and to the identification of novel biomarkers for prognosis and diagnosis (3). Herein, we review key studies using these approaches, their major findings, and the challenges ahead.
Edible seaweeds extracts: Characterization and functional properties for health conditions
Abstract
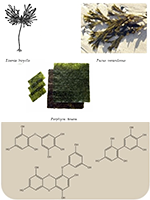
Seaweeds are popular foods due to claimed beneficial health effects, but for many there is a lack of scientific evidence. In this study, extracts of the edible seaweeds Aramé, Nori, and Fucus are compared. Our approach intends to clarify similarities and differences in the health properties of these seaweeds, thus contributing to target potential applications for each. Additionally, although Aramé and Fucus seaweeds are highly explored, information on Nori composition and bioactivities is scarce. The aqueous extracts of the seaweeds were obtained by decoction, then fractionated and characterized according to their composition and biological activity. It was recognized that fractioning the extracts led to bioactivity reduction, suggesting a loss of bioactive compounds synergies. The Aramé extract showed the highest antioxidant activity and Nori exhibited the highest potential for acetylcholinesterase inhibition. The identification of the bioactive compounds in the extracts allowed to see that these contained a mixture of phloroglucinol polymers, and it was suggested that Nori’s effect on acetylcholinesterase inhibition may be associated with a smaller sized phlorotannins capable of entering the enzyme active site. Overall, these results suggest a promising potential for the use of these seaweed extracts, mainly Aramé and Nori, in health improvement and management of diseases, namely those associated to oxidative stress and neurodegeneration.
Absence of EPAC1 signaling to stabilize CFTR in intestinal organoids
Abstract
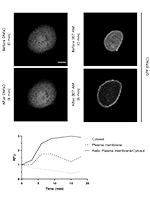
The plasma membrane (PM) stability of the cystic fibrosis transmembrane conductance regulator (CFTR), the protein which when mutated causes Cystic Fibrosis (CF), relies on multiple interaction partners that connect CFTR to signaling pathways, including cAMP signaling. It was previously shown that activation of exchange protein directly activated by cAMP 1 (EPAC1) by cAMP promotes an increase in CFTR PM levels in airway epithelial cells. However, the relevance of this pathway in other tissues, particularly the intestinal tissue, remains uncharacterized. Here, we used Western blot and forskolin-induced swelling assay to demonstrate that the EPAC1 protein is not expressed in the intestinal organoid model, and consequently the EPAC1 stabilization pathway is not in place. On the other hand, using cell surface biotinylation, EPAC1-mediated stabilization of PM CFTR is observed in intestinal cell lines. These results indicate that the EPAC1 stabilization pathway also occurs in intestinal cells and is a potential target for the development of novel combinatorial therapies for treatment of CF.
A drug screening reveals minocycline hydrochloride as a therapeutic option to prevent breast cancer cells extravasation across the blood-brain barrier
Abstract
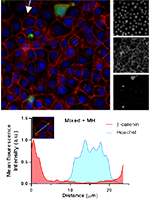
Among breast cancer (BC) patients, 15–25% develop BC brain metastases (BCBM), a severe condition due to the limited therapeutic options, which points to the need for preventive strategies. We aimed to find a drug able to boost blood–brain barrier (BBB) properties and prevent BC cells (BCCs) extravasation, among PI3K, HSP90, and EGFR inhibitors and approved drugs. We used BCCs (4T1) and BBB endothelial cells (b.End5) to identify molecules with toxicity to 4T1 cells and safe for b.End5 cells. Moreover, we used those cells in mixed cultures to perform a high-throughput microscopy screening of drugs’ ability to ameliorate BBB properties and prevent BCCs adhesion and migration across the endothelium, as well as to analyse miRNAs expression and release profiles. KW-2478, buparlisib, and minocycline hydrochloride (MH) promoted maximal expression of the junctional protein β-catenin and induced 4T1 cells nucleus changes. Buparlisib and MH further decreased 4T1 adhesion. MH was the most promising in preventing 4T1 migration and BBB disruption, tumour and endothelial cytoskeleton-associated proteins modifications, and miRNA deregulation. Our data revealed MH’s ability to improve BBB properties, while compromising BCCs viability and interaction with BBB endothelial cells, besides restoring miRNAs’ homeostasis, paving the way for MH repurposing for BCBM prevention.
Searching for a Paradigm Shift in Auger-Electron Cancer Therapy with Tumor-Specific Radiopeptides Targeting the Mitochondria and/or the Cell Nucleus
Abstract
Although 99mTc is not an ideal Auger electron (AE) emitter for Targeted Radionuclide Therapy (TRT) due to its relatively low Auger electron yield, it can be considered a readily available “model” radionuclide useful to validate the design of new classes of AE-emitting radioconjugates. With this in mind, we performed a detailed study of the radiobiological effects and mechanisms of cell death induced by the dual-targeted radioconjugates 99mTc-TPP-BBN and 99mTc-AO-BBN (TPP = triphenylphosphonium; AO = acridine orange; BBN = bombesin derivative) in human prostate cancer PC3 cells. 99mTc-TPP-BBN and 99mTc-AO-BBN caused a remarkably high reduction of the survival of PC3 cells when compared with the single-targeted congener 99mTc-BBN, leading to an augmented formation of γH2AX foci and micronuclei. 99mTc-TPP-BBN also caused a reduction of the mtDNA copy number, although it enhanced the ATP production by PC3 cells. These differences can be attributed to the augmented uptake of 99mTc-TPP-BBN in the mitochondria and enhanced uptake of 99mTc-AO-BBN in the nucleus, allowing the irradiation of these radiosensitive organelles with the short path-length AEs emitted by 99mTc. In particular, the results obtained for 99mTc-TPP-BBN reinforce the relevance of targeting the mitochondria to promote stronger radiobiological effects by AE-emitting radioconjugates.
Exploring YAP1-centred networks linking dysfunctional CFTR to epithelial-mesenchymal transition
Abstract
Mutations in the CFTR anion channel cause Cystic Fibrosis (CF) and have also been related to higher cancer incidence. Previously we proposed that this is linked to an emerging role of functional CFTR in protecting against epithelial-mesenchymal transition (EMT). However, the pathways bridging dysfunctional CFTR to EMT remain elusive. Here, we applied systems biology to address this question. Our data show that YAP1 is aberrantly active in the presence of mutant CFTR, interacting with F508del- but not with wt-CFTR, and that YAP1 knockdown rescues F508del-CFTR processing and function. Subsequent analysis of YAP1 interactors and roles in cells expressing either wt- or F508del-CFTR, reveal that YAP1 is an important mediator of the fibrotic/EMT processes in CF. Alongside, five main pathways emerge here as key in linking mutant CFTR to EMT, namely: 1) the Hippo pathway; 2) the Wnt pathway; 3) the TGFβ pathway; 4) the p53 pathway; and 5) MYC signaling. Several potential hub proteins which mediate the crosstalk among these pathways were also identified, appearing as potential therapeutic targets for both CF and cancer.
CFTR interactome mapping using the mammalian membrane two-hybrid high-throughput screening system
Abstract

Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) is a chloride and bicarbonate channel in secretory epithelia with a critical role in maintaining fluid homeostasis. Mutations in CFTR are associated with Cystic Fibrosis (CF), the most common lethal autosomal recessive disorder in Caucasians. While remarkable treatment advances have been made recently in the form of modulator drugs directly rescuing CFTR dysfunction, there is still considerable scope for improvement of therapeutic effectiveness. Here, we report the application of a high-throughput screening variant of the Mammalian Membrane Two-Hybrid (MaMTH-HTS) to map the protein–protein interactions of wild-type (wt) and mutant CFTR (F508del), in an effort to better understand CF cellular effects and identify new drug targets for patient-specific treatments. Combined with functional validation in multiple disease models, we have uncovered candidate proteins with potential roles in CFTR function/CF pathophysiology, including Fibrinogen Like 2 (FGL2), which we demonstrate in patient-derived intestinal organoids has a significant effect on CFTR functional expression.
Systems approaches to unravel molecular function: High-content siRNA screen identifies TMEM16A traffic regulators as potential drug targets for Cystic Fibrosis
Abstract
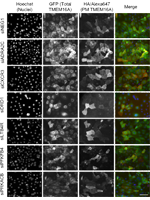
An attractive approach to treat people with Cystic Fibrosis (CF), a life-shortening disease caused by mutant CFTR, is to compensate for the absence of this chloride/bicarbonate channel by activating alternative (non-CFTR) chloride channels. One obvious target for such “mutation-agnostic” therapeutic approach is TMEM16A (anoctamin-1/ANO1), a calcium-activated chloride channel (CaCC) which is also expressed in the airways of people with CF, albeit at low levels. To find novel TMEM16A regulators of both traffic and function, with the main goal of identifying candidate CF drug targets, we performed a fluorescence cell-based high-throughput siRNA microscopy screen for TMEM16A trafficking using a double-tagged construct expressed in human airway cells. About 700 genes were screened (2 siRNAs per gene) of which 262 were identified as candidate TMEM16A modulators (179 siRNAs enhanced and 83 decreased TMEM16A traffic), being G-protein coupled receptors (GPCRs) enriched on the primary hit list. Among the 179 TMEM16A traffic enhancer siRNAs subjected to secondary screening 20 were functionally validated. Further hit validation revealed that siRNAs targeting two GPCRs – ADRA2C and CXCR3 – increased TMEM16A-mediated chloride secretion in human airway cells, while their overexpression strongly diminished calcium-activated chloride currents in the same cell model. The knockdown, and likely also the inhibition, of these two TMEM16A modulators is therefore an attractive potential therapeutic strategy to increase chloride secretion in CF.
An open-source high-content analysis workflow for CFTR function measurements using the forskolin-induced swelling assay
Abstract
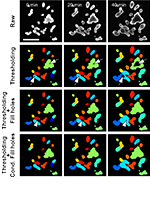
Motivation
The forskolin-induced swelling (FIS) assay has become the preferential assay to predict the efficacy of approved and investigational CFTR-modulating drugs for individuals with cystic fibrosis (CF). Currently, no standardized quantification method of FIS data exists thereby hampering inter-laboratory reproducibility.
Results
We developed a complete open-source workflow for standardized high-content analysis of CFTR function measurements in intestinal organoids using raw microscopy images as input. The workflow includes tools for (i) file and metadata handling; (ii) image quantification and (iii) statistical analysis. Our workflow reproduced results generated by published proprietary analysis protocols and enables standardized CFTR function measurements in CF organoids.
Availability and implementation
All workflow components are open-source and freely available: the htmrenamer R package for file handling https://github.com/hmbotelho/htmrenamer; CellProfiler and ImageJ analysis scripts/pipelines https://github.com/hmbotelho/FIS_image_analysis; the Organoid Analyst application for statistical analysis https://github.com/hmbotelho/organoid_analyst; detailed usage instructions and a demonstration dataset https://github.com/hmbotelho/FIS_analysis. Distributed under GPL v3.0.
Organoids as a Personalized Medicine Tool for Ultra-Rare Mutations in Cystic Fibrosis: the Case of S955P and 1717-2A>G
Abstract
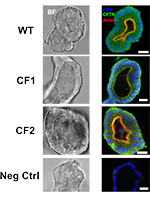
Background
For most of the >2000 CFTR gene variants reported, neither the associated disease liability nor the underlying basic defect are known, and yet these are essential for disease prognosis and CFTR-based therapeutics. Here we aimed to characterize two ultra-rare mutations - 1717-2A > G (c.1585-2A > G) and S955P (p.Ser955Pro) - as case studies for personalized medicine.
Methods
Patient-derived rectal biopsies and intestinal organoids from two individuals with each of these mutations and F508del (p.Phe508del) in the other allele were used to assess CFTR function, response to modulators and RNA splicing pattern. In parallel, we used cellular models to further characterize S955P independently of F508del and to assess its response to CFTR modulators.
Results
Results in both rectal biopsies and intestinal organoids from both patients evidence residual CFTR function. Further characterization shows that 1717-2A > G leads to alternative splicing generating <1% normal CFTR mRNA and that S955P affects CFTR gating. Finally, studies in organoids predict that both patients are responders to VX-770 alone and even more to VX-770 combined with VX-809 or VX-661, although to different levels.
Conclusion
This study demonstrates the high potential of personalized medicine through theranostics to extend the label of approved drugs to patients with rare mutations.
Full Rescue of F508del-CFTR Processing and Function by CFTR Modulators Can Be Achieved by Removal of Two Regulatory Regions
Abstract
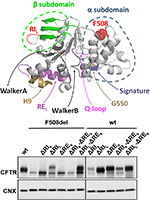
Cystic Fibrosis (CF) is caused by mutations in the CF Transmembrane conductance Regulator (CFTR), the only ATP-binding cassette (ABC) transporter functioning as a channel. Unique to CFTR is a regulatory domain which includes a highly conformationally dynamic region—the regulatory extension (RE). The first nucleotide-binding domain of CFTR contains another dynamic region—regulatory insertion (RI). Removal of RI rescues the trafficking defect of CFTR with F508del, the most common CF-causing mutation. Here we aimed to assess the impact of RE removal (with/without RI or genetic revertants) on F508del-CFTR trafficking and how CFTR modulator drugs VX-809/lumacaftor and VX-770/ivacaftor rescue these variants. We generated cell lines expressing DRE and DRI CFTR (with/without genetic revertants) and assessed CFTR expression, stability, plasma membrane levels, and channel activity. Our data demonstrated that DRI significantly enhanced rescue of F508del-CFTR by VX-809. While the presence of the RI seems to be precluding full rescue of F508del-CFTR processing by VX-809, this region appears essential to rescue its function by VX-770, suggesting some contradictory role in rescue of F508del-CFTR by these two modulators. This negative impact of RI removal on VX-770-stimulated currents on F508del-CFTR can be compensated by deletion of the RE which also leads to the stabilization of this mutant. Despite both regions being conformationally dynamic, RI precludes F508del-CFTR processing while RE affects mostly its stability and channel opening.
Abstract
Mutations associated with cystic fibrosis (CF) have complex effects on the cystic fibrosis transmembrane conductance regulator (CFTR) protein. The most common CF mutation, F508del, disrupts the processing to and stability at the plasma membrane and function as a Cl- channel. CFTR is surrounded by a dynamic network of interacting components, referred to as the CFTR Functional Landscape, that impact its synthesis, folding, stability, trafficking and function. CFTR interacting proteins can be manipulated by functional genomic approaches to rescue the trafficking and functional defects characteristic of CF. Here we review recent efforts to elucidate the impact of genetic variation on the ability of the nascent CFTR polypeptide to interact with the proteostatic environment. We also provide an overview of how specific components of this protein network can be modulated to rescue the trafficking and functional defects associated with the F508del variant of CFTR. The identification of novel proteins playing key roles in the processing of CFTR could pave the way for their use as novel therapeutic targets to provide synergistic correction of mutant CFTR for the greater benefit of individuals with CF.
Folding Status Is Determinant Over Traffic-Competence in Defining CFTR Interactors in the Endoplasmic Reticulum
Abstract
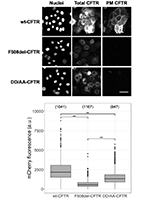
The most common cystic fibrosis-causing mutation (F508del, present in ~85% of CF patients) leads to CFTR misfolding, which is recognized by the endoplasmic reticulum (ER) quality control (ERQC), resulting in ER retention and early degradation. It is known that CFTR exit from the ER is mediated by specific retention/sorting signals that include four arginine-framed tripeptide (AFT) retention motifs and a diacidic (DAD) exit code that controls the interaction with the COPII machinery. Here, we aim at obtaining a global view of the protein interactors that regulate CFTR exit from the ER. We used mass spectrometry-based interaction proteomics and bioinformatics analyses to identify and characterize proteins interacting with selected CFTR peptide motifs or full-length CFTR variants retained or bypassing these ERQC checkpoints. We conclude that these ERQC trafficking checkpoints rely on fundamental players in the secretory pathway, detecting key components of the protein folding machinery associated with the AFT recognition and of the trafficking machinery recognizing the diacidic code. Furthermore, a greater similarity in terms of interacting proteins is observed for variants sharing the same folding defect over those reaching the same cellular location, evidencing that folding status is dominant over ER escape in shaping the CFTR interactome.
Unravelling the antitumoral potential of novel bis(thiosemicarbazonato) Zn(II) complexes: structural and cellular studies
Abstract
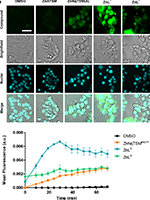
Background
The development of pharmacologically active compounds based on bis(thiosemicarbazones) (BTSC) and on their coordination to metal centers constitutes a promising field of research. We have recently explored this class of ligands and their Cu(II) complexes for the design of cancer theranostics agents with enhanced uptake by tumoral cells. In the present work, we expand our focus to aliphatic and aromatic BTSC Zn(II) complexes bearing piperidine/morpholine pendant arms. The new complexes ZnL1–ZnL4 were characterized by a variety of analytical techniques, which included single-crystal X-ray crystallography for ZnL2 and ZnL3. Taking advantage of the fluorescent properties of the aromatic complexes, we investigated their cellular uptake kinetics and subcellular localization. Furthermore, we tried to elucidate the mechanism of action of the cytotoxic effect observed in human cancer cell line models. The results show that the aliphatic complexes (ZnL1 and ZnL2) have a symmetrical structure, while the aromatic counterparts (ZnL3 and ZnL4) have an asymmetrical nature. The cytotoxic activity was higher for the aromatic BTSC complexes, as well as the cellular uptake, evaluated by measurement of intracellular Zn accumulation. Among the most active complexes, ZnL3 presented the fastest uptake kinetics and lysosomal localization assessed by live-cell microscopy. Detailed studies of its impact on cellular production of reactive oxygen species and impairment of lysosomal membrane integrity reinforced the influence of the pendant piperidine in the biological performance of aromatic BTSC Zn(II) complexes.
R560S: A class II CFTR mutation that is not rescued by current modulators
Abstract
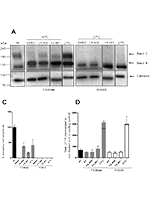
Background
New therapies modulating defective CFTR have started to hit the clinic and others are in trial or under development. The endeavour of drug discovery for CFTR protein rescue is however difficult one since over 2000 mutations have been reported. For most of these, especially the rare ones, the associated defects, the respective functional class and their responsiveness to available modulators are still unknown. Our aim here was to characterize the rare R560S mutation using patient-derived materials (rectal biopsies and intestinal organoids) from one CF individual homozygous for this mutation, in parallel with cellular models expressing R560S-CFTR and to assess the functional and biochemical responses to CFTR modulators.
Methods
Intestinal organoids were prepared from rectal biopsies and analysed by RT-PCR (to assess CFTR mRNA), by Western blot (to assess CFTR protein) and by forskolin-induced swelling (FIS) assay. A novel cell line expressing R560S-CFTR was generated by stably transducing the CFBE parental cell line and used to assess R560S-CFTR processing and function. Both intestinal organoids and the cellular model were used to assess efficacy of CFTR modulators in rescuing this mutation.
Results
Our results show that: R560S does not affect CFTR mRNA splicing; R560S affects CFTR protein processing, totally abrogating the production of its mature form; R560S-CFTR evidences no function as a Cl− channel; and none of the modulators tested rescued R560S-CFTR processing or function.
Conclusion
Altogether, these results indicate that R560S is a class II mutation. However, unlike F508del, it cannot be rescued by any of the CFTR modulators tested.
The neuronal S100B protein is a calcium-tuned suppressor of amyloid-β aggregation
Abstract

Amyloid-β (Aβ) aggregation and neuroinflammation are consistent features in Alzheimer’s disease (AD) and strong candidates for the initiation of neurodegeneration. S100B is one of the most abundant proinflammatory proteins that is chronically up-regulated in AD and is found associated with senile plaques. This recognized biomarker for brain distress may, thus, play roles in amyloid aggregation which remain to be determined. We report a novel role for the neuronal S100B protein as suppressor of Aβ42 aggregation and toxicity. We determined the structural details of the interaction between monomeric Aβ42 and S100B, which is favored by calcium binding to S100B, possibly involving conformational switching of disordered Aβ42 into an α-helical conformer, which locks aggregation. From nuclear magnetic resonance experiments, we show that this dynamic interaction occurs at a promiscuous peptide-binding region within the interfacial cleft of the S100B homodimer. This physical interaction is coupled to a functional role in the inhibition of Aβ42 aggregation and toxicity and is tuned by calcium binding to S100B. S100B delays the onset of Aβ42 aggregation by interacting with Aβ42 monomers inhibiting primary nucleation, and the calcium-bound state substantially affects secondary nucleation by inhibiting fibril surface–catalyzed reactions through S100B binding to growing Aβ42 oligomers and fibrils. S100B protects cells from Aβ42-mediated toxicity, rescuing cell viability and decreasing apoptosis induced by Aβ42 in cell cultures. Together, our findings suggest that molecular targeting of S100B could be translated into development of novel approaches to ameliorate AD neurodegeneration.
Full rescue of F508del-CFTR processing and function by CFTR modulators can be achieved by removal of two unique regulatory regions
Abstract

Background and Purpose
Cystic Fibrosis (CF) is caused by mutations in the CF Transmembrane conductance Regulator (CFTR), the only ABC transporter functioning as a channel. Unique to CFTR are two highly conformationally dynamic regions: the regulatory extension (RE) and regulatory insertion (RI). Removal of the latter rescues the trafficking defect of CFTR with F508del, the most common CF-causing mutation. We aimed here to assess the impact of RE removal (alone or with RI or genetic revertants) on F508del-CFTR traffic and how CFTR modulator drugs corrector VX-809/lumacaftor and potentiator VX-770/ivacaftor rescue these combined variants so as to gain insight into the mechanism of action (MoA) of these drugs.
Experimental Approach
We generated ΔRE and ΔRI CFTR variants (with and without genetic revertants) by site-directed mutagenesis and used them to stably transfect BHK cell lines. We studied CFTR expression and stability by Western blotting and pulse-chase respectively, plasma membrane levels by cell surface biotinylation and channel activity by the iodide efflux technique.
Key Results
Our data demonstrate that ΔRI significantly enhanced rescue of F508del-CFTR by VX-809. Thus, while the presence of the regulatory insertion seems to be precluding full rescue of F508del-CFTR processing by VX-809, this region appears essential to rescue its function by VX-770, thus suggesting some contradictory role in rescue of F508del-CFTR by these two modulators. Nevertheless, this negative impact of RI removal on VX-770-stimulated currents on F508del-CFTR can be compensated by deletion of the regulatory extension which also leads to the stabilization of this mutant. We thus propose that, despite both these regions being conformationally active, RI precludes F508del-CFTR processing while RE affects mostly its stability and channel opening.
A novel microscopy-based assay identifies extended synaptotagmin-1 (ESYT1) as a positive regulator of anoctamin 1 traffic
Abstract
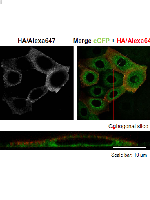
An attractive possibility to treat Cystic Fibrosis (CF), a severe condition caused by dysfunctional CFTR, an epithelial anion channel, is through the activation of alternative (non-CFTR) anion channels. Anoctamin 1 (ANO1) was demonstrated to be a Ca2+-activated chloride channel (CaCC) and thus of high potential to replace CFTR. Despite that ANO1 is expressed in human lung CF tissue, it is present at the cell surface at very low levels. In addition, little is known about regulation of ANO1 traffic, namely which factors promote its plasma membrane (PM) localization. Here, we generated a novel cellular model, expressing an inducible 3HA-ANO1-eGFP construct, and validated its usage as a microscopy tool to monitor for ANO1 traffic. We demonstrate the robustness and specificity of this cell-based assay, by the identification of siRNAs acting both as ANO1 traffic enhancer and inhibitor, targeting respectively COPB1 and ESYT1 (extended synaptotagmin-1), the latter involved in coupling of the endoplasmic reticulum to the PM at specific microdomains. We further show that knockdown of ESYT1 (and family members ESYT2 and ESYT3) significantly decreased ANO1 current density. This ANO1 cell-based assay constitutes an important tool to be further used in high-throughput screens and drug discovery of high relevance for CF and cancer.
Investigating Alternative Transport of Integral Plasma Membrane Proteins from the ER to the Golgi: Lessons from the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR)
Abstract

Secretory traffic became a topical field because many important cell regulators are plasma membrane proteins (transporters, channels, receptors), being thus key targets in biomedicine and drug discovery. Cystic fibrosis (CF), caused by defects in a single gene encoding the CF transmembrane conductance regulator (CFTR), constitutes the most common of rare diseases and certainly a paradigmatic one.Here we focus on five different approaches that allow biochemical and cellular characterization of CFTR from its co-translational insertion into the ER membrane to its delivery to the plasma membrane.
Correction of a Cystic Fibrosis Splicing Mutation by Antisense Oligonucleotides
Abstract
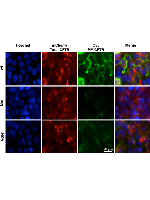
Cystic fibrosis (CF), the most common life-threatening genetic disease in Caucasians, is caused by ~2,000 different mutations in the CF transmembrane conductance regulator (CFTR) gene. A significant fraction of these (~13%) affect pre-mRNA splicing for which novel therapies have been somewhat neglected. We have previously described the effect of the CFTR splicing mutation c.2657+5G>A in IVS16, showing that it originates transcripts lacking exon 16 as well as wild-type transcripts. Here, we tested an RNA-based antisense oligonucleotide (AON) strategy to correct the aberrant splicing caused by this mutation. Two AONs (AON1/2) complementary to the pre-mRNA IVS16 mutant region were designed and their effect on splicing was assessed at the RNA and protein levels, on intracellular protein localization and function. To this end, we used the 2657+5G>A mutant CFTR minigene stably expressed in HEK293 Flp-In cells that express a single copy of the transgene. RNA data from AON1-treated mutant cells show that exon 16 inclusion was almost completely restored (to 95%), also resulting in increased levels of correctly localized CFTR protein at the plasma membrane (PM) and with increased function. A novel two-color CFTR splicing reporter minigene developed here allowed the quantitative monitoring of splicing by automated microscopy localization of CFTR at the PM. The AON strategy is thus a promising therapeutic approach for the specific correction of alternative splicing.
Transcriptome meta-analysis reveals common differential and global gene expression profiles in cystic fibrosis and other respiratory disorders and identifies CFTR regulators
Abstract
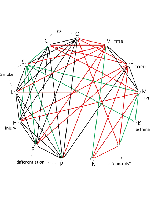
A meta-analysis of 13 independent microarray data sets was performed and gene expression profiles from cystic fibrosis (CF), similar disorders (COPD: chronic obstructive pulmonary disease, IPF: idiopathic pulmonary fibrosis, asthma), environmental conditions (smoking, epithelial injury), related cellular processes (epithelial differentiation/regeneration), and non-respiratory “control” conditions (schizophrenia, dieting), were compared. Similarity among differentially expressed (DE) gene lists was assessed using a permutation test, and a clustergram was constructed, identifying common gene markers. Global gene expression values were standardized using a novel approach, revealing that similarities between independent data sets run deeper than shared DE genes. Correlation of gene expression values identified putative gene regulators of the CF transmembrane conductance regulator (CFTR) gene, of potential therapeutic significance. Our study provides a novel perspective on CF epithelial gene expression in the context of other lung disorders and conditions, and highlights the contribution of differentiation/EMT and injury to gene signatures of respiratory disease.
Protein Traffic Disorders: an Effective High-Throughput Fluorescence Microscopy Pipeline for Drug Discovery
Abstract
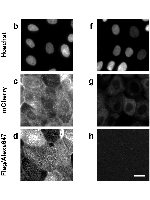
Plasma membrane proteins are essential molecules in the cell which mediate interactions with the exterior milieu, thus representing key drug targets for present pharma. Not surprisingly, protein traffic disorders include a large range of diseases sharing the common mechanism of failure in the respective protein to reach the plasma membrane. However, specific therapies for these diseases are remarkably lacking. Herein, we report a robust platform for drug discovery applied to a paradigmatic genetic disorder affecting intracellular trafficking - Cystic Fibrosis. This platform includes (i) two original respiratory epithelial cellular models incorporating an inducible double-tagged traffic reporter; (ii) a plasma membrane protein traffic assay for high-throughput microscopy screening; and (iii) open-source image analysis software to quantify plasma membrane protein traffic. By allowing direct scoring of compounds rescuing the basic traffic defect, this platform enables an effective drug development pipeline, which can be promptly adapted to any traffic disorder-associated protein and leverage therapy development efforts.
Structural Heterogeneity and Bioimaging of S100 Amyloid Assemblies
Abstract
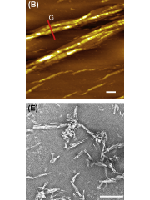
S100 proteins are small EF-hand Ca2+-binding proteins involved in diverse cellular processes such as cell survival, proliferation and differentiation. Many S100 proteins also bind additional metal ions which modulate their folding and activity. The expression levels of many S100 proteins are significantly increased in cancer, neurodegenerative, inflammatory and autoimmune diseases. Recently, S100A8/A9 were identified as major amyloidogenic proteins in corpora amylacea inclusions of prostate cancer patients and found to undergo metal-mediated amyloid oligomerization and fibrillation in vitro. The amyloidogenic potential has also been found in other S100 family members, suggesting that amyloid-like assemblies may play an important role in S100 biologic activity in health and disease. In this chapter we address the structural diversity of S100 aggregates, as revealed by high-resolution bioimaging techniques. We start by overviewing the structural diversity of individual S100 proteins and their functional oligomers; this is followed by analysis of their amyloidogenic aggregation potential, general characterization of S100 amyloids, and the role of metal ions in aggregation pathways. The morphologic diversity of the aggregates formed by different types of S100 protein is illustrated by electron and atomic force microscopy data. The chapter ends by overviewing the amyloid formation by S100A8/A9 in the aging prostate, showing how microscopy techniques can be used to characterize in vitro and ex vivo amyloids.
Intrinsically Disordered and Aggregation Prone Regions Underlie β-Aggregation in S100 Proteins
Abstract

Plasma membrane proteins are essential molecules in the cell which mediate interactions with the exterior milieu, thus representing key drug targets for present pharma. Not surprisingly, protein traffic disorders include a large range of diseases sharing the common mechanism of failure in the respective protein to reach the plasma membrane. However, specific therapies for these diseases are remarkably lacking. Herein, we report a robust platform for drug discovery applied to a paradigmatic genetic disorder affecting intracellular trafficking - Cystic Fibrosis. This platform includes (i) two original respiratory epithelial cellular models incorporating an inducible double-tagged traffic reporter; (ii) a plasma membrane protein traffic assay for high-throughput microscopy screening; and (iii) open-source image analysis software to quantify plasma membrane protein traffic. By allowing direct scoring of compounds rescuing the basic traffic defect, this platform enables an effective drug development pipeline, which can be promptly adapted to any traffic disorder-associated protein and leverage therapy development efforts.
Biochemical and Biophysical Characterization of Recombinant Yeast Proteasome Maturation Factor Ump1
Abstract
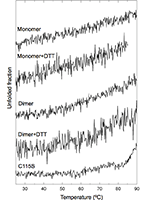
Protein degradation is essential for maintaining cellular homeostasis. The proteasome is the central enzyme responsible for non-lysosomal protein degradation in eukaryotic cells. Although proteasome assembly is not yet completely understood, a number of cofactors required for proper assembly and maturation have been identified. Ump is a short-lived maturation factor required for the efficient biogenesis of the 20S proteasome. Upon the association of the two precursor complexes, Ump is encased and is rapidly degraded after the proteolytic sites in the interior of the nascent proteasome are activated. In order to further understand the mechanisms behind proteasomal maturation, we expressed and purified yeast Ump in E. coli for biophysical and structural analysis. We show that recombinant Ump is purified as a mixture of different oligomeric species and that oligomerization is mediated by intermolecular disulfide bond formation involving the only cysteine residue present in the protein. Furthermore, a combination of bioinformatic, biochemical and structural analysis revealed that Ump shows characteristics of an intrinsically disordered protein, which might become structured only upon interaction with the proteasome subunits.
S100A6 Amyloid Fibril Formation Is Calcium-modulated and Enhances Superoxide Dismutase-1 (SOD1) Aggregation
Abstract
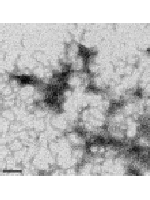
S100A6 is a small EF-hand calcium- and zinc-binding protein involved in the regulation of cell proliferation and cytoskeletal dynamics. It is overexpressed in neurodegenerative disorders and a proposed marker for Amyotrophic Lateral Sclerosis (ALS). Following recent reports of amyloid formation by S100 proteins, we investigated the aggregation properties of S100A6. Computational analysis using aggregation predictors Waltz and Zyggregator revealed increased propensity within S100A6 helices HI and HIV. Subsequent analysis of Thioflavin-T binding kinetics under acidic conditions elicited a very fast process with no lag phase and extensive formation of aggregates and stacked fibrils as observed by electron microscopy. Ca2+ exerted an inhibitory effect on the aggregation kinetics, which could be reverted upon chelation. An FT-IR investigation of the early conformational changes occurring under these conditions showed that Ca2+ promotes anti-parallel β-sheet conformations that repress fibrillation. At pH 7, Ca2+ rendered the fibril formation kinetics slower: time-resolved imaging showed that fibril formation is highly suppressed, with aggregates forming instead. In the absence of metals an extensive network of fibrils is formed. S100A6 oligomers, but not fibrils, were found to be cytotoxic, decreasing cell viability by up to 40%. This effect was not observed when the aggregates were formed in the presence of Ca2+. Interestingly, native S1006 seeds SOD1 aggregation, shortening its nucleation process. This suggests a cross-talk between these two proteins involved in ALS. Overall, these results put forward novel roles for S100 proteins, whose metal-modulated aggregation propensity may be a key aspect in their physiology and function.
Metal ions as modulators of protein conformation and misfolding in neurodegeneration
*equally contributing authors
Abstract
Protein misfolding and conformational changes are a cornerstone of neurodegenerative diseases involving formation and deposition of toxic protein oligomers. Although mutations favor protein aggregation, physiological factors such as labile metal ions within the cellular environment are likely to play a role. Metal ions such as calcium, zinc and copper are key players in brain neurobiology, their homeostasis is altered in most neurodegenerative conditions and they are found within proteinaceous inclusions from patients. In this review we will elucidate the intricate interplay between protein (mis)folding and metal ions, discussing how metals modulate protein folding and influence protein energetics, with specific attention on conformational changes and structural fluctuations. In particular, the influence of metal ion dyshomeostasis during neurodegeneration and the effects of the unique physical and chemical properties at the synaptic environment will be discussed in the context of protein deposition. These interactions will be illustrated by specific examples of proteins involved in neurodegenerative diseases including α-synuclein, tau, superoxide dismutase 1, the prion protein and the amyloid-β peptide. With this approach we aim to systematize the effects of metal ions on protein conformers and illustrate pathways through which they modulate protein aggregation, under different conceptual mechanisms that bridge protein structure, metallochemistry and neurobiology.
The Sulfur Oxygenase Reductase from the Mesophilic Bacterium Halothiobacillus neapolitanus Is a Highly Active Thermozyme
Abstract
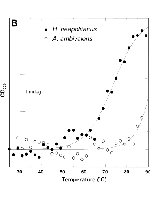
A biochemical, biophysical, and phylogenetic study of the sulfur oxygenase reductase (SOR) from the mesophilic gammaproteobacterium Halothiobacillus neapolitanus (HnSOR) was performed in order to determine the structural and biochemical properties of the enzyme. SOR proteins from 14 predominantly chemolithoautotrophic bacterial and archaeal species are currently available in public databases. Sequence alignment and phylogenetic analysis showed that they form a coherent protein family. The HnSOR purified from Escherichia coli after heterologous gene expression had a temperature range of activity of 10 to 99°C with an optimum at 80°C (42 U/mg protein). Sulfite, thiosulfate, and hydrogen sulfide were formed at various stoichiometries in a range between pH 5.4 and 11 (optimum pH 8.4). Circular dichroism (CD) spectroscopy and dynamic light scattering showed that the HnSOR adopts secondary and quaternary structures similar to those of the 24-subunit enzyme from the hyperthermophile Acidianus ambivalens (AaSOR). The melting point of the HnSOR was ≈20°C lower than that of the AaSOR, when analyzed with CD-monitored thermal unfolding. Homology modeling showed that the secondary structure elements of single subunits are conserved. Subtle changes in the pores of the outer shell and increased flexibility might contribute to activity at low temperature. We concluded that the thermostability was the result of a rigid protein core together with the stabilizing effect of the 24-subunit hollow sphere.
Analysis of S100 oligomers and amyloids
Abstract
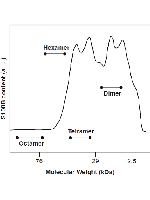
The S100 proteins are a large family of 10–12 kDa EF-hand signaling proteins that bind calcium, and in some cases zinc and copper, functioning as central regulators in a diversity of cellular processes. These proteins have tissue, cell, and subcellular-specific expression patterns, and many have an extracellular function. Altogether, these properties underlie their functional diversity and involvement in several pathological conditions including cancer, inflammation, and neurodegeneration. S100 proteins exhibit considerable structural plasticity, being able to exist as monomers or assemble into dimers, higher oligomers, and amyloids, frequently in a metal-dependent manner. Many of these oligomers are functionally relevant, and S100 amyloids have been recently found in prostatic inclusions. Here, we report experimental procedures for the isolation and quantitation of S100 oligomers from tissues, purification of recombinant human S100 protein for assays and use as standards, and an amyloidogenesis assay that allows monitoring the formation of S100 β-oligomers and amyloids in apo- and metal-bound S100 proteins.
Structural reorganization renders enhanced metalloprotein stability
Abstract
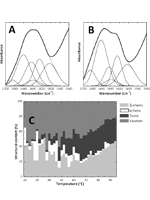
The enhanced stability of a mesophilic metalloprotein was assessed using biophysical spectroscopies. Significant local structural interconversions during thermal insult account for a reorganization of the protein scaffold, without disturbing the active metal site. This cushioning mechanism is proposed to be a generic property of metalloproteins contributing to enhanced stability.
Metal ions and protein folding: conformational and functional interplay
Abstract
Metal ions are cofactors in about 30% of all proteins, where they fulfill catalytical and structural roles. Due to their unique chemistry and coordination properties they effectively expand the intrinsic polypeptide properties (by participating in catalysis or electron transfer reactions), stabilize protein conformations (like in zinc fingers) and mediate signal transduction (by promoting functionally relevant protein conformational changes). However, metal ions can also exert have deleterious effects in living systems by incorporating in non-native binding sites, promoting aberrant protein aggregation or mediating redox cycling with generation of reactive oxygen and nitrogen species. For this reason, the characterization of the roles of metal ions as modulators of protein conformation and stability provides fundamental knowledge on protein folding properties and is instrumental in establishing the molecular basis of disease. In this thesis we have analyzed protein folding processes using model protein systems incorporating covalently bound metal cofactors – iron-sulfur (FeS) proteins – or where metal ion binding is reversible and associated conformational readjustments – the S100 proteins.(...)
Natural and amyloid self-assembly of S100 proteins: structural basis of functional diversity
Abstract
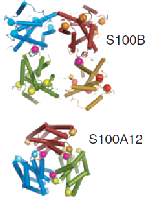
The S100 proteins are 10–12 kDa EF-hand proteins that act as central regulators in a multitude of cellular processes including cell survival, proliferation, differentiation and motility. Consequently, many S100 proteins are implicated and display marked changes in their expression levels in many types of cancer, neurodegenerative disorders, inflammatory and autoimmune diseases. The structure and function of S100 proteins are modulated by metal ions via Ca2+ binding through EF-hand motifs and binding of Zn2+ and Cu2+ at additional sites, usually at the homodimer interfaces. Ca2+ binding modulates S100 conformational opening and thus promotes and affects the interaction with p53, the receptor for advanced glycation endproducts and Toll-like receptor 4, among many others. Structural plasticity also occurs at the quaternary level, where several S100 proteins self-assemble into multiple oligomeric states, many being functionally relevant. Recently, we have found that the S100A8/A9 proteins are involved in amyloidogenic processes in corpora amylacea of prostate cancer patients, and undergo metal-mediated amyloid oligomerization and fibrillation in vitro. Here we review the unique chemical and structural properties of S100 proteins that underlie the conformational changes resulting in their oligomerization upon metal ion binding and ultimately in functional control. The possibility that S100 proteins have intrinsic amyloid-forming capacity is also addressed, as well as the hypothesis that amyloid self-assemblies may, under particular physiological conditions, affect the S100 functions within the cellular milieu.
Role of a novel disulfide bridge within the all-beta fold of soluble Rieske proteins
Abstract
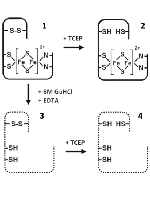
Rieske proteins and Rieske ferredoxins are present in the three domains of life and are involved in a variety of cellular processes. Despite their functional diversity, these small Fe–S proteins contain a highly conserved all-β fold, which harbors a [2Fe–2S] Rieske center. We have identified a novel subtype of Rieske ferredoxins present in hyperthermophilic archaea, in which a two-cysteine conserved SKTPCX(2–3)C motif is found at the C-terminus. We establish that in the Acidianus ambivalens representative, Rieske ferredoxin 2 (RFd2), these cysteines form a novel disulfide bond within the Rieske fold, which can be selectively broken under mild reducing conditions insufficient to reduce the [2Fe–2S] cluster or affect the secondary structure of the protein, as shown by visible circular dichroism, absorption, and attenuated total reflection Fourier transform IR spectroscopies. RFd2 presents all the EPR, visible absorption, and visible circular dichroism spectroscopic features of the [2Fe–2S] Rieske center. The cluster has a redox potential of +48 mV (25 °C and pH 7) and a pKa of 10.1 ± 0.2. These shift to +77 mV and 8.9 ± 0.3, respectively, upon reduction of the disulfide. RFd2 has a melting temperature near the boiling point of water (Tm = 99 °C, pH 7.0), but it becomes destabilized upon disulfide reduction (ΔTm = −9 °C, ΔCm = −0.7 M guanidinium hydrochloride). This example illustrates how the incorporation of an additional structural element such as a disulfide bond in a highly conserved fold such as that of the Rieske domain may fine-tune the protein for a particular function or for increased stability.
Metal ions modulate the folding and stability of the tumor suppressor protein S100A2
Abstract

The EF-hand protein S100A2 is a cell cycle regulator involved in tumorigenesis, acting through regulation of the p53 activation state. Metal ion-free S100A2 is homodimeric and contains two Ca2+-binding sites and two Zn2+-binding sites per subunit, whereby the Zn2+ ion binding to one of the sites is coordinated by residues from two homodimers. The effect of selective binding of these metal ions was investigated using site-specific mutants which lacked one or both zinc sites. CD analysis of secondary structure changes on metallation showed that Zn2+ binding was associated with a decrease in the secondary structure content, whereas Ca2+ had the opposite effect in two of the three S100A2 mutants studied. The energy of unfolding (ΔGU) of the apo wild-type S100A2 was determined to be 89.9 kJ·mol−1, and the apparent midpoint transition temperature (Tmapp) was 58.4 °C. In addition, a detailed study of the urea and thermal unfolding of the S100A2 mutants in different metallation states (apo, Zn2+ and Ca2+) was performed. Thermal denaturation experiments showed that Zn2+ acts as a destabilizer and Ca2+ as a stabilizer of the protein conformation. This suggests a synergistic effect between metal binding, protein stability and S100A2 biological activity, according to which Ca2+ activates and stabilizes the protein, the opposite being observed on Zn2+ binding.
A proteomic approach toward the selection of proteins with enhanced intrinsic conformational stability
Abstract

A detailed understanding of the molecular basis of protein folding and stability determinants partly relies on the study of proteins with enhanced conformational stability properties, such as those from thermophilic organisms. In this study, we set up a methodology aiming at identifying the subset of cytosolic hyperstable proteins using Sulfurispharea sp., a hyperthermophilic archaeon, able to grow between 70 and 97 degrees C, as a model organism. We have thermally and chemically perturbed the cytosolic proteome as a function of time (up to 96 h incubation at 90 degrees C), and proceeded with analysis of the remaining proteins by combining one- and two-dimensional gel electrophoresis, liquid chromatography fractionation, and protein identification by N-terminal sequencing and mass spectrometry methods. In total, 14 proteins with enhanced stabilities which are involved in key cellular processes such as detoxification, nucleic acid processing, and energy metabolism were identified including a superoxide dismutase, a peroxiredoxin, and a ferredoxin. We demonstrate that these proteins are biologically active after extensive thermal treatment of the proteome. The relevance of these and other targets is discussed in terms of the organism's ecology. This work thus illustrates an experimental approach aimed at mining a proteome for hyperstable proteins, a valuable tool for target selection in protein stability and structural studies.